- Home
- »
- Metal Finishing
Precision Metal Finishing & Surface Treatment Services | XML
Looking for a one-stop solution to enhance the durability, conductivity, or aesthetics of your machined parts? XML offers a comprehensive suite of metal finishing services. From advanced anodizing and electroplating to precision polishing, we ensure your components meet the most rigorous industrial standards. As an all-around hardware processing manufacturer, we integrate CNC machining with premium surface treatments to deliver market-ready products. Explore our factory capabilities below.
Certifications : ISO 9001:2015 | AS9100D | ITAR Registered
Anodizing
The oxide film formed on the alloy in a natural environment is only 0.01 to 0.1 μm thick, providing insufficient protection and failing to meet the requirements of engineering applications for corrosion resistance, wear resistance, and aesthetic appeal. Anodizing is currently the most widely used surface hardening technique for aluminum alloys. It involves the formation of an aluminum oxide (Al₂O₃) layer on the aluminum substrate through an electrochemical reaction. This layer bonds firmly to the substrate, possesses high hardness, and, due to its porous structure, can undergo coloring and sealing treatments.


Mechanism of the Anodizing Reaction
In the anodizing process, an aluminum workpiece is placed in an electrolyte solution as the anode, and a direct current is applied. Two reactions occur simultaneously on the anode surface:
Film-forming reaction: Aluminum combines with oxygen evolved at the anode to form Al₂O₃;
Dissolution reaction: The electrolyte (sulfuric acid, for example) dissolves the generated Al₂O₃.
| Process Type | Electrolyte | Coating Thickness | Characteristics | Typical Applications |
| Chromic Acid Anodizing (CAA) | 3% – 10% Chromic Acid | 5 – 8μm | Thin coating; maintains workpiece precision; does not change dimensions. | Precision parts, welded assemblies, aerospace structural components. |
| Sulfuric Acid Anodizing (SAA) | 10% – 20% Sulfuric Acid | 8 – 25μm | High transparency; low cost; dyeable (colorable). | Consumer electronics, building materials, daily necessities. |
| Hard Anodizing (Hardcoat) | Sulfuric Acid + Organic Acid | 25 – 150μm | High hardness (HV300 – 600); wear-resistant; insulating. | Cylinders, hydraulic components, military equipment. |
| Parameter | Recommended Range | Limit Range | Consequences of Deviation |
Sulfuric Acid Concentration | 15% – 20% | 10% – 25% | Low concentration leads to thin coating; high concentration causes rapid dissolution and loose coating structure. |
| Aluminum Ion ($Al^{3+}$) Concentration | 5 – 15g/L | < 20g/L | Exceeding 20g/L causes the coating to turn gray and decreases corrosion resistance. |
| Bath Temperature | 18 – 22°C | 15 – 25°C | > 25°C: Loose coating or powdering; < 15°C: Dense coating but increased brittleness. |
| Current Density | 1.2 – 1.5A/dm² | 0.8 – 2.0A/dm² | Too high causes burning/scorching; too low results in thin coating and low production efficiency. |
| Anodizing Time | 30 – 60min | Based on thickness requirement | Insufficient time leads to inadequate thickness; excessive time expands pore diameter and reduces adhesion. |
| Chloride ($Cl^-$) Concentration | ≤ 25mg/L | ≤ 50mg/L | Exceeding the limit leads to pitting corrosion and perforation. |
Pre-treatment Process
The quality of the pre-treatment process directly affects the uniformity, adhesion, and appearance of the anodized coating. Defects introduced by incomplete or improper pre-treatment cannot be repaired after anodizing.
Raw Material Acceptance Criteria
Aluminum alloys of different grades should not be mixed during loading. Series 2 (aluminum-copper), Series 7 (aluminum-zinc), Series 5 (aluminum-magnesium), and Series 6 (aluminum-magnesium-silicon) alloys exhibit significant differences in oxidation rates under the same electrolytic parameters; processing them in the same tank can easily lead to selective oxidation and color variations in the coating.
Extrusion marks, scratches, and original corrosion pits are difficult to completely eliminate during pretreatment, and these defects will become magnified after oxidation. Surface quality should be controlled during the machining stage.
Mechanical Finishing
Sandblasting: Removes scale and casting skin to achieve a uniform sandblasted finish. The surface is highly reactive after sandblasting; the anodizing process must begin within 4 hours to prevent secondary oxidation or contamination.
Grinding/Polishing: If the alkaline etching time during pretreatment is insufficient after mirror polishing, residual stress layers or dense oxide films may cause mottling in the anodized coating. Polishing compounds must be thoroughly degreased.
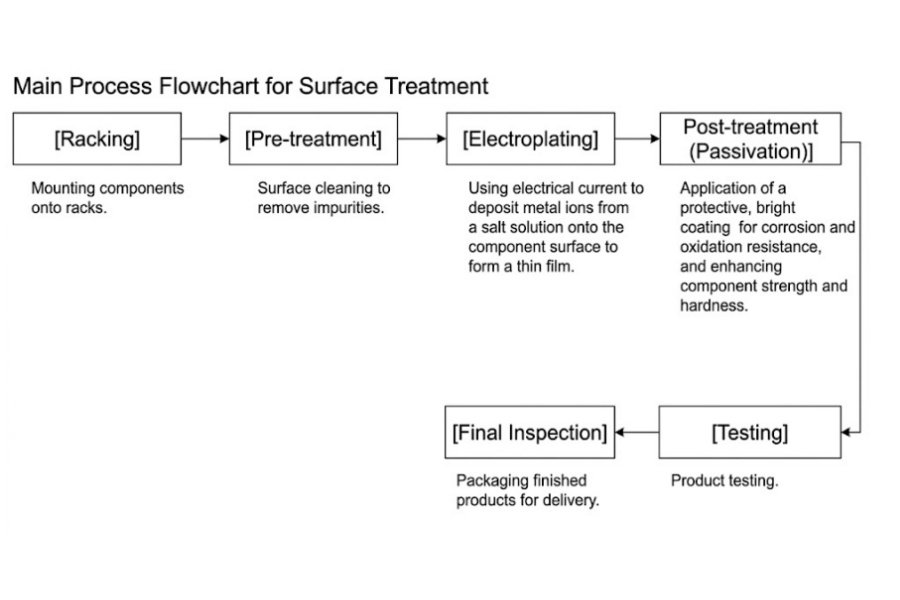
Electroplating
Surface treatment generally refers to a process that modifies the surface of a workpiece to enhance its appearance or performance. At EagleP, surface treatment typically refers to electroplating, which is one of the most widely used surface treatment technologies, offering a variety of metal coatings and high cost-effectiveness. Our surface treatment services primarily include electroplating, as well as a limited range of electroless plating services.


Electroplating is a process that alters the surface properties of a metal part by depositing a layer of metal onto its surface using an electric current. First, the metal part to be coated is immersed in a solution containing the metal to be deposited. The metal ions in the electroplating solution carry a positive charge and are attracted to the negatively charged metal part.
Our electroplating services can be categorized into functional electroplating and decorative electroplating. Functional electroplating processes improve the electrical conductivity, wear resistance, and corrosion resistance of components, making them essential for the components’ functionality. Decorative electroplating processes are primarily used for aesthetic purposes. We also offer electroless plating, which applies corrosion-resistant materials and other functional materials to the surface of objects to enhance their corrosion resistance, wear resistance, and decorative properties.
Common Polishing Methods and How They Work

Mechanical Polishing
Mechanical polishing is a method that removes protrusions from the surface of a workpiece through cutting or plastic deformation to achieve a smooth finish. It typically involves the use of oilstone bars, wool wheels, sandpaper, and other tools, and is primarily performed manually. For applications requiring high surface quality, superfinishing can be employed. Superfinishing involves pressing a specially designed tool against the workpiece surface while it rotates at high speed in a polishing slurry containing abrasive particles. This technique can achieve a surface roughness of Ra 0.008 μm, which is the finest surface finish achievable among all polishing methods; it is commonly used for optical lens molds. Mechanical polishing is the primary method for mold polishing.
Chemical Polishing
Chemical polishing works by using substances in a chemical medium to selectively dissolve microscopic protrusions on the surface rather than depressions, thereby producing a smooth finish. This method is suitable for polishing workpieces with complex shapes and allows for the simultaneous polishing of multiple workpieces, making it highly efficient. The surface roughness achieved through chemical polishing is typically Ra 10 μm.


Electrolytic Polishing
The basic principle of electrolytic polishing is the same as that of chemical polishing: it smooths the surface by selectively dissolving microscopic protrusions on the material’s surface. Compared to chemical polishing, it eliminates the effects of cathodic reactions, resulting in superior results.
Ultrasonic Polishing
Ultrasonic polishing is a machining method that utilizes ultrasonic vibrations generated by the cross-section of a tool to polish brittle and hard materials using an abrasive suspension. The workpiece is placed in the abrasive suspension and positioned within an ultrasonic field; the oscillations of the ultrasonic waves cause the abrasives to grind and polish the surface of the workpiece. Ultrasonic machining exerts relatively low macroscopic forces and does not cause deformation of the workpiece; however, the fabrication and installation of the tooling are relatively difficult.

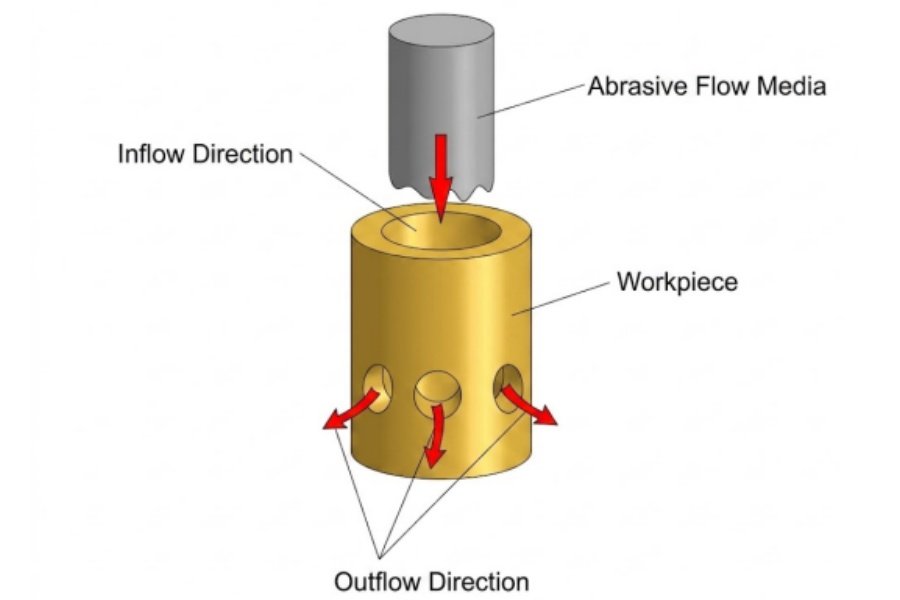
Fluid Polishing
Fluid polishing relies on flowing liquid and the abrasive particles it carries to scour the surface of the workpiece, thereby achieving a polished finish. Hydrodynamic grinding is hydraulically driven, and the medium consists primarily of a special compound (a polymer-based substance) with good flow properties at low pressure, mixed with abrasive particles; the abrasive can be silicon carbide powder.
Magnetic Grinding and Polishing
Magnetic grinding and polishing utilizes magnetic abrasive particles to form an abrasive brush under the influence of a magnetic field, thereby grinding the workpiece. This method offers high processing efficiency, excellent quality, and easily controllable processing conditions. With the selection of appropriate abrasives, surface roughness can reach Ra 0.1 μm.

Electrocoating technology
component of modern coating processes, has been widely adopted in industries such as automotive and home appliances due to its unique advantages. This article aims to summarize the fundamentals of electrocoating technology and the characteristics of its equipment, while refining and polishing the relevant content to help readers better understand and apply this technology.
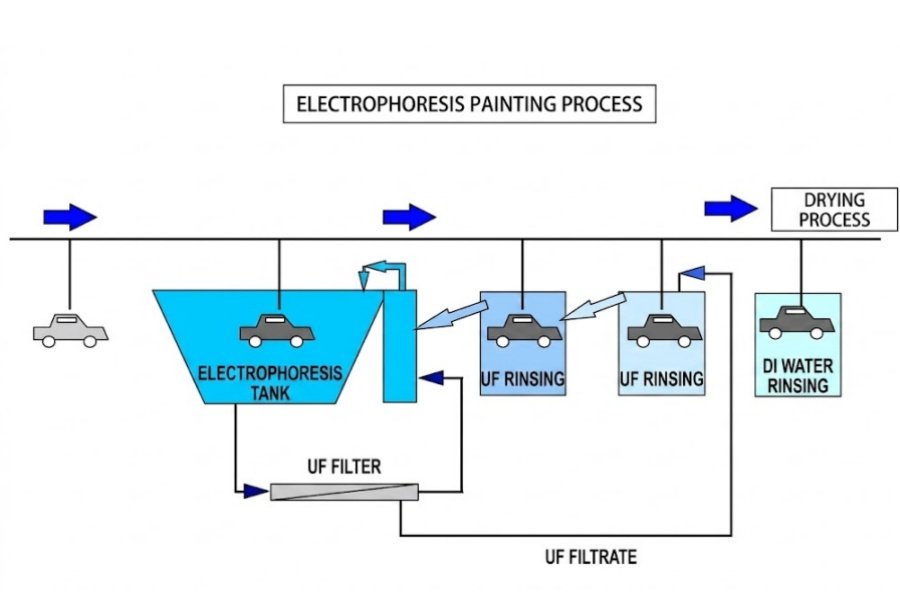
This is a process in which charged paint particles are caused by an electric field to deposit onto the surface of the substrate, forming a uniform coating. Depending on the charge carried by the paint particles, electrophoretic coating can be classified into anionic electrophoresis and cationic electrophoresis. Since the discovery of the electrophoretic phenomenon, electrophoretic coating technology has continued to evolve. From the initial practical application of anionic electrophoresis, to the subsequent practical application of cationic electrophoresis, and finally to the emergence of new-generation and eco-friendly electrophoretic coating technologies, the application scope and performance of electrophoretic coating technology have continuously expanded and improved.
Electrocoating technology offers numerous advantages. First, the coating conditions are easy to control and manage, enabling uniform film thickness, high penetration, and rust prevention on inner panels. Second, because it uses water-based coatings, electrophoretic coating poses virtually no risks of fire or solvent hazards, resulting in a low environmental impact. Furthermore, the solid content in electrophoretic coatings helps prevent defects such as clumping, paint defects, and sagging. However, electrophoretic coating also has some limitations, such as the requirement that the substrate be electrically conductive, the difficulty in changing coatings and colors, and the need to prevent contamination from impurities such as ions and oil.
Painting and Powder Coating in Coating Production Linese
Surface treatment is a critical process for enhancing product appearance and improving corrosion resistance and wear resistance. As the two primary surface coating technologies in coating production lines, painting and powder coating each offer unique advantages and have distinct applications.

Painting Process on Coating Production Lines: The painting process utilizes a wide variety of coatings, such as fluorocarbon paint, acrylic paint, epoxy resin paint, and polyurethane paint. These paints contain solvents and curing agents that are mixed to form a liquid, which is then atomized by a spray gun and evenly applied to the surface of the workpiece. The liquid coating process offers a rich variety of colors and high gloss, capable of meeting diverse aesthetic requirements.
Powder Coating Process on Coating Production Lines: Powder coating technology utilizes powder coatings (commonly known as plastic powders), also referred to as electrostatic powder coating. The primary types include epoxy-polyester powder coatings and epoxy powder coatings. Powder coatings exist in a solid state and do not require solvents or curing agents; they can be applied directly to the workpiece through electrostatic adhesion or mechanical spraying. Powder coatings offer environmental benefits; their 100% solids content means no solvent emissions, thereby reducing environmental pollution and safety hazards.
| Comparison Dimension | Liquid Spray Painting | Powder Coating |
| Physical Form | Liquid (contains resin, pigment, solvent/water, additives) | Solid (dry powder, contains resin, pigment, curing agent) |
| Coating Thickness | Thinner, typically 15 – 50 μm per layer | Thicker, typically 60 – 120 μm |
| Impact on Tolerance | Minor impact; suitable for precision machined surfaces requiring strict dimensional accuracy. | Significant impact; strict masking is usually required for precision holes, threads, or assembly surfaces. |
| Curing Temperature | Air-dries at room temperature, or low-temperature baking (typically 60°C – 80°C). | High-temperature baking for melting and cross-linking (typically 160°C – 220°C for 15-20 minutes). |
| Surface Physical Properties | Average wear and scratch resistance; moderate adhesion. | Excellent wear and impact resistance; strong adhesion, tough coating. |
| Corrosion / Weather Resistance | Average rust and corrosion resistance (unless using special anti-corrosion paint); extremely high maximum color and gloss limits. | Outstanding corrosion, acid, and alkali resistance; good edge coverage, less prone to rusting at sharp corners. |
| Appearance Defect Control | Highly dependent on operator skill; prone to sagging, bubbling, and uneven thickness. | Electrostatic adsorption ensures uniform powder distribution, no sagging, and less prone to pinholes. |
| Environmental & Compliance | Mostly contains VOCs (Volatile Organic Compounds), requiring complex exhaust treatment; water-based paints are more eco-friendly but face high compliance costs in US/EU markets. | Near-zero VOC emissions, overspray powder is recyclable (up to 95%+ utilization), easily complies with strict international environmental standards like RoHS. |
| Material & Cost | Low equipment investment; however, low coating utilization (approx. 30% – 50%) and higher manual maintenance costs. | High investment for automated production lines; however, low per-unit material cost, suitable for large-volume, scale production. |
| Material Suitability | Extremely wide range of applications, including metal, plastic, wood, and non-standard materials that cannot withstand high temperatures. | Primarily suitable for conductive metals (steel, aluminum, iron, etc.), and materials must be able to withstand temperatures around 200°C without deforming. |
Metal Finishing
Anodizing enhances corrosion resistance and allows for vibrant color finishes on aluminum parts.
Electroplating adds a thin, protective metal coating to improve durability and appearance.
Polishing smooths the surface to achieve a shiny, mirror-like finish for enhanced aesthetics 
Powder coating provides a durable, colored finish that resists chipping, scratching, and fading.
Electrophoretic coating provides a smooth, bonded protective layer, improving durability, aesthetic appeal.
Metal Finishing - Frequently Asked Questions (FAQ)
At XML Tech, we provide high-quality Metal Finishing to enhance the durability and appearance of your precision parts. Whether you need Anodizing for wear resistance or Electroplating for conductivity, our cost-effective processes ensure your components meet strict industrial standards with superior protection and a professional finish.
Metal finishing is a post-machining process used to enhance the properties of a workpiece. Its primary purposes are to improve corrosion resistance, increase surface hardness (wear resistance), enhance electrical conductivity, or provide a specific aesthetic finish (such as color or texture).
It depends on your base material and requirements:
Anodizing is primarily used for Aluminum alloys. It creates an integrated oxide layer that is excellent for wear resistance and color dyeing.
Electroplating is more versatile and can be applied to Steel, Copper, and Aluminum. It is preferred when you need a specific metal layer (like Nickel, Chrome, or Zinc) for conductivity or high-performance corrosion protection.
Chromic Acid Anodizing (CAA) is the best choice for high-precision parts. It produces a very thin coating (5–8μm) that does not significantly change the dimensions of the part, making it ideal for aerospace and tight-tolerance assemblies.
Most aluminum parts treated with Sulfuric Acid Anodizing (SAA) can be dyed in a wide range of colors (Black, Blue, Red, Gold, etc.). However, Hard Anodizing usually results in a naturally dark grey or bronze finish, which is more difficult to dye but offers superior hardness.

